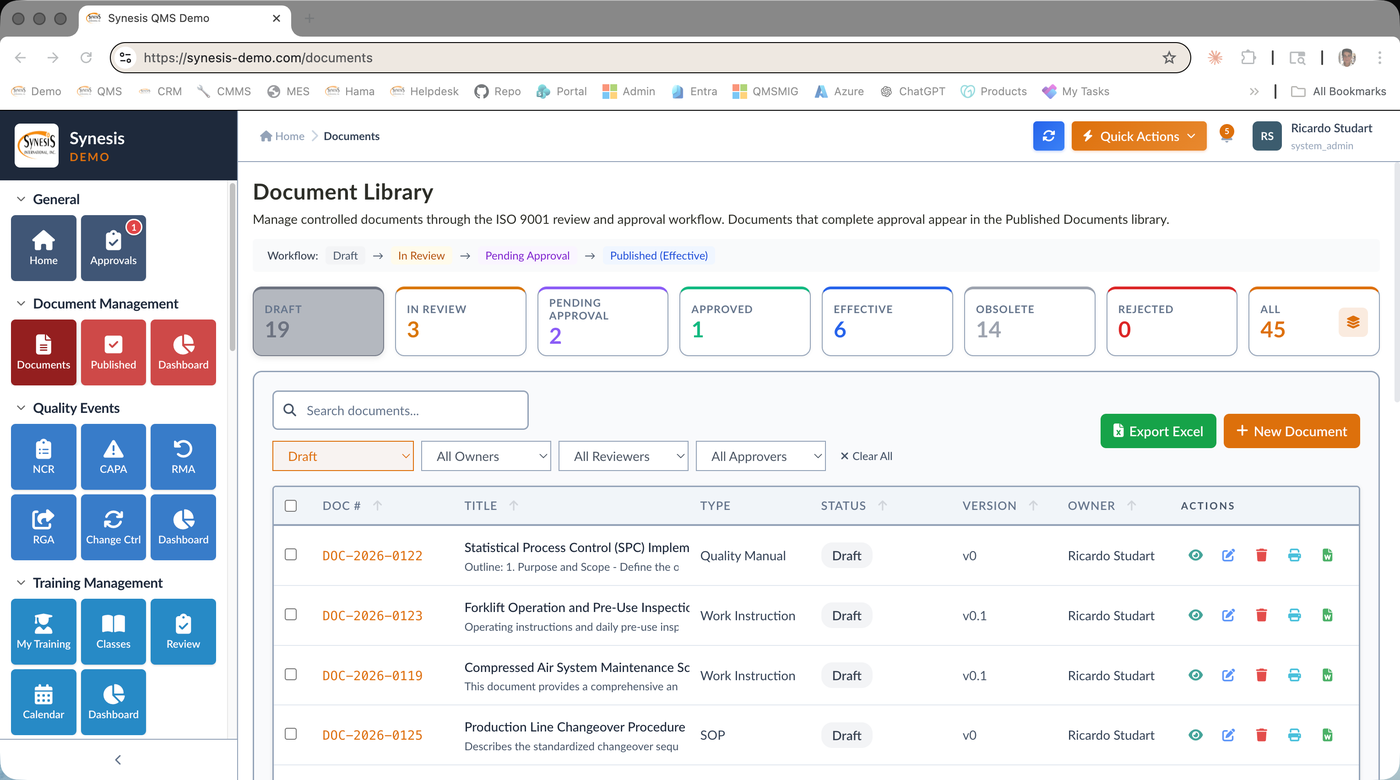
Controlled Document Library
Single Source of Truth
All controlled documents — SOPs, Work Instructions, Forms, Specifications, Policies, Manuals, Procedures, Record Templates, External Documents — managed in one library with type, owner, reviewer, approver, and retention period.